S. pombe Genome-wide Heterozygous Deletion Mutant Screening Service

BIONEER의 약물작용점 규명 서비스: GPScreen™-FAST
약물 작용점을 규명하는 혁신 기술로서, 분열 효모 게놈 적중 라이브러리를 이용하여 유전체 수준에서 신약 후보물질들의 약물 작용점을 빠르고 정확하게 규명해 드립니다.
GPScreen™-FAST 기술의 특장점
-
분열효모 게놈 적중 라이브러리 기반의 세계 유일의 혁신적인 약물 작용점 규명 기술
-
유전체를 기반으로 거의 모든 유형의 약물 타겟 스크리닝 가능
-
살아있는 세포 기반의 약효 평가
-
NGS 기반 DNA barcode를 분석하여 빠르고 정확한 스크리닝
-
약물 작용점, 독성 평가, 의약재창출, 천연물 작용 기전 규명 등에 적용 가능
GPScreen™-FAST 기술의 응용 분야
-
약물 작용점 규명 및 독성 평가
-
우수화합물의 선별
-
의약 재창출 및 약효 개선
-
천연물의 타겟 발굴 및 작용 기전 연구
-
화학 유전체 분석
개요
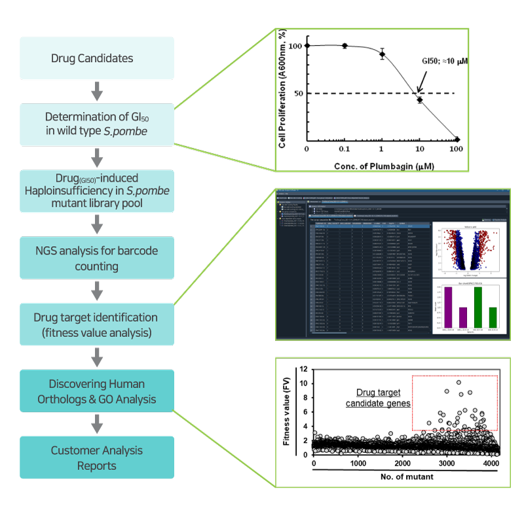
정확한 약물 작용점 규명은 신약개발의 성공률 증대를 위한 핵심 이슈로서, 신약 후보 물질의 작용 기전 (mode-of-action)의 이해 뿐만 아니라 약효 개선 및 부작용 문제를 분자 수준에서 해결하기 위한 신약 개발의 첫 단계입니다. 그리고 바이오니아는 인간과 효모 유전체 기반에서 약물의 타겟을 제시하는 기술인 GPScreen™-FAST를 개발하였습니다. 본 기술은 인간 생명현상 연구를 위한 주요 모델인 분열효모 (S.pombe) 게놈 적중 라이브러리를 이용하여, 효모 유전체 수준에서 약물의 작용점을 탐색하는 시스템으로 약물 작용점 규명, 유전자 발현 및 synthetic lethal 등과 같은 유전자 기능분석 및 약물 스크리닝에 사용될 수 있습니다. 또한 주요 질환인 암, 대사질환 뿐만 아니라 그간 개발에 소외된 희귀병까지 거의 모든 질환 영역의 신약 후보 물질들의 작용점 규명에 적용 가능합니다. 이를 통해 궁극적으로 신약 개발 성공률을 획기적으로 개선해 드릴 것입니다.
GPScreen™-FAST은 바이오니아, 한국 생명공학연구원 (KRIBB) 및 영국 왕립 암연구소(Cancer Research UK)의 공동 연구를 통해 개발된 세계 유일의 분열효모 (S. pombe) 게놈 적중 라이브러리 (Kim et al. Nat. Biotech, 28, 617–623 (2010)를 기반으로 하고 있습니다. 바이오니아는 이 라이브러리에 대한 모든 사업 독점권을 보유하고 있으며 유전자 기능 분석, 약물 작용점 규명 및 세포 기능 연구를 위해 제공하고 있습니다.
게놈적중라이브러리는 분열효모 전체 게놈의 약 98.5% 유전자 변이체들로 구성되어 있습니다. 개별 변이체들은 상동 유전자 재조합 (homologous recombination)에 의해 정상 세포에 존재하는 한 쌍의 개별 유전자들 중 하나가 결손되어 있으며, 각 개별 유전자 변이체들은 고유의 DNA barcode로 tagging 되어 있습니다 (아래 그림 참조). 그리고 균일하게 성장한 전체 개별 변이체들을 pool로 제작하였으며, pool에 존재하는 각 DNA barcode들을 빠르고 정확하게 NGS 기술로 분석합니다.
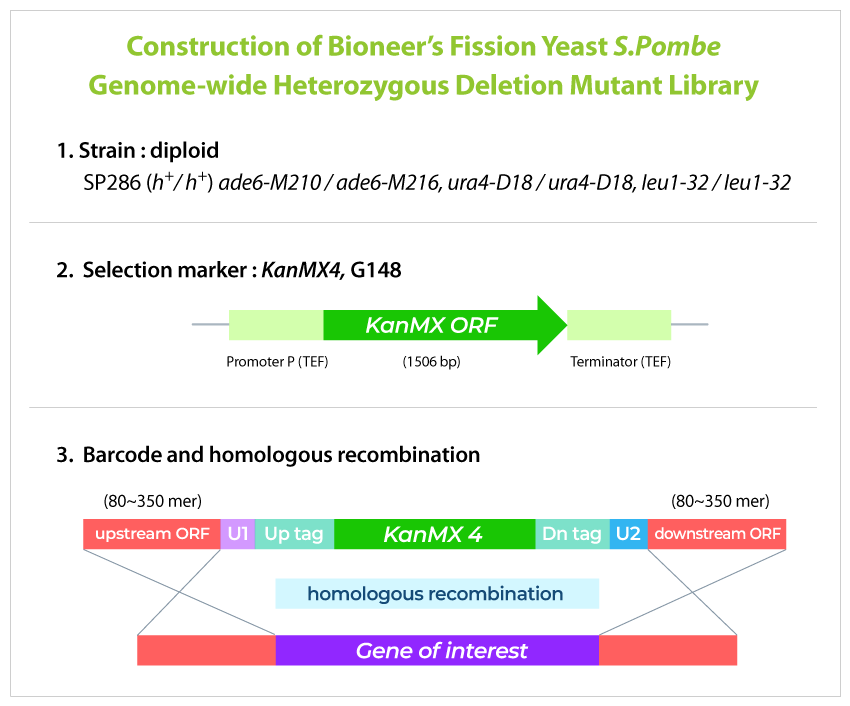
GPScreen™-FAST 원리
'Drug-induced Haploinsufficiency (DIH)'는 heterozygous deletion mutant(특정 단백질이 정상세포에 비해 절반만 발현되는 변이체)에서 약물의 민감도가 증가하는 현상을 의미합니다. 이러한 현상은 약물이 타겟으로 하는 특정한 유전자가 결손된 변이체에서 나타나며, 약물 작용점 규명의 효과적인 수단으로 여겨지고 있습니다. 그간 많은 다양한 유형의 약물들에 대한 작용점이 이러한 원리를 이용하여 발아 효모 (S. cerevisiae) 게놈 적중 라이브러리를 통해 제시 된 바 있습니다 (Baetz K et al., 2004; Lum et al., 2004).발아 효모 대비 분열 효묘에만 존재하는 인간과 유사한 생물학적 특징으로 인해 분열 효모는 보다 이상적인 세포 모델로 여겨지고 있습니다 (Vyas A et al., 2021).
GPScreen™-FAST는 유전자 결손 변이체들의 약물유래 민감도 확인을 위해 고유 DNA barcode count를 NGS 기술로 빠르고 정확하게 분석합니다. 또한 GPScreen Analysis Software (GPSAS)를 활용하여, NGS data로부터 각 유전자들의 fitness value (FV) 값을 분석하고 최종적으로 약물의 타겟 유전자를 빠르고 정확하게 탐색할 수 있도록 합니다.
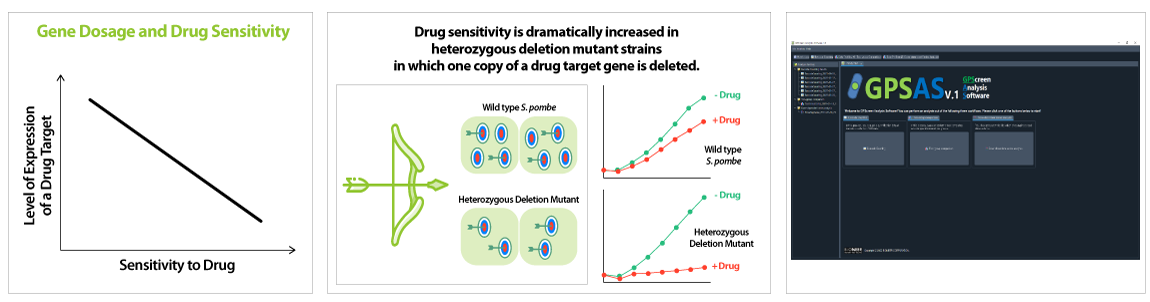
GPScreen™-FAST 성능
GPScreen™-FAST 기술은 유전체 수준에서 약물의 작용점을 정확하게 제시할 수 있습니다.
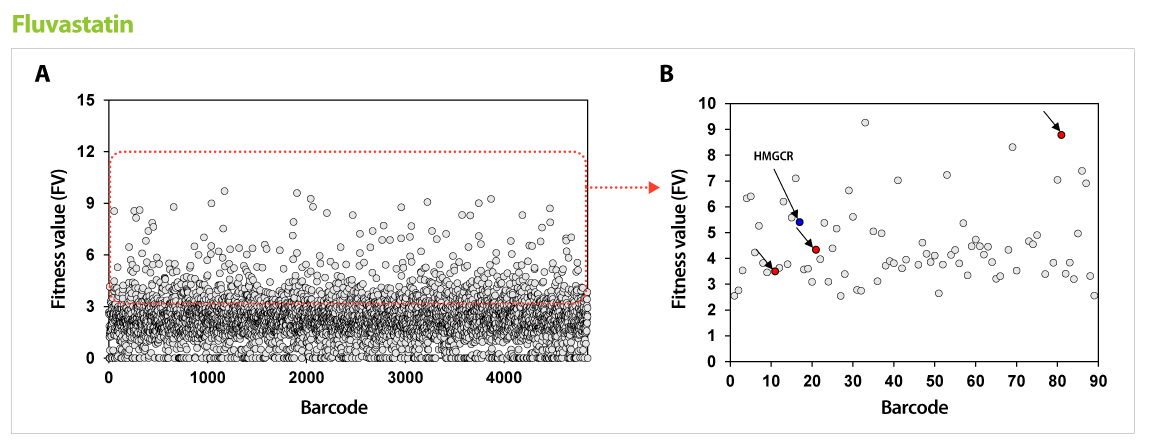
Figure 1. Results of GPScreen™-FAST to discover candidate target genes of Fluvastatin. (A) Barcode counts by NGS analysis. Barcode number of total mutant strains (pool) was read by NGS analysis. (B) Candidate target genes of Fluvastatin. Among selected candidate genes from GPScreenTM-FAST (red boundary in (A)), drug-induced haploinsufficiency (DIH) assay in individual mutant strain was performed as a secondary screening. As a results, known-target gene (HMGCR, blue circle) and expected-target genes (red circle) were detected.
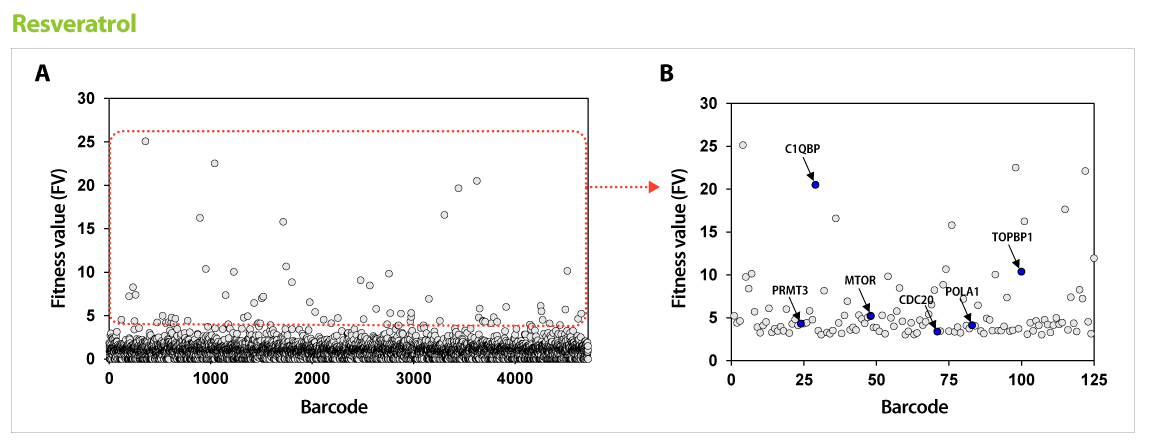
Figure 2. Results of GPScreen™-FAST to discover candidate target genes of Resveratrol. (A) Barcode counts by NGS analysis. Barcode number of total mutant strains (pool) was read by NGS analysis. (B) Candidate target genes of Resveratrol. Among selected candidate genes from GPScreenTM-FAST (red boundary in (A)), drug-induced haploinsufficiency (DIH) assay in individual mutant strain was performed as a secondary screening. As a results, known-target genes (blue circle) were discovered.


▶ GPScreen을 통해 발굴된 약물 작용점을 이용하여 인체 세포에서 타겟 검증 연구가 가능합니다.
I인체세포에서의 약물 타겟 검증 방법
바이오니아는 약 2만여 종의 인체 유전자에 대한 siRNA 라이브러리를 보유하고 있으며,
발굴된 타겟의 human ortholog를 이용하여 인체 세포에서의 유전자 발현 억제를 통한 타겟을 검증할 수 있습니다.
1) 인체 세포에서 약물에 의한 인체 상동 유전자 발현 조절 분석
Figure 1. Sunitinib 이 Hela 인간 세포주에서 인간 상동 유전자인 POLG의 발현을 억제시키는 것을 확인
(A) 인체 세포에서의 sunitinib 효과를 확인하기 위해 인간 자궁경부암 세포주인 Hela 세포에 sunitinib을 처리한 후 POLG 단백질의 발현량을 확인했습니다. POLG 항체와 goat anti-rabbit IgG-TRITC 항체를 이용하여 세포면역염색 후 컨포컬 현광현미경에서 관찰했습니다.
(B) Hela 세포에 sunitinib을 처리한 후 POLG mRNA 양을 qRT-PCR로 측정했습니다.
2) 바이오니아의 human siRNA 라이브러리를 이용하여 인간상동유전자 발현 억제 효과 분석
Figure 2. 인간 자궁경부암 세포주인 Hela 세포에서 POLG 발현 억제 시 sunitinib 에 의한 세포 독성이 현격히 증가
(upper panel) Hela 세포주에 POLG 타겟 siRNA(5 nM, 48h)를 처리한 후 세포에 sunitinib (5 µM, 48h) 을 처리하여 SRB assay로 세포 양을 측정하였습니다.
(bottom panel) SRB assay 한 세포를 광학 현미경으로 관찰하였습니다.
IIGPScreen™ 기술의 신약개발 후보 도출에의 적용
추가로, GPScreen™ 기술은 신약 개발 초기 단계에서 약물 후보들 간의 우수 화합물 선별에도 이용 가능합니다. 본 기술을 이용한 유전체 수준에서의 타겟 프로파일을 분석 시 독성 약물은 비독성 약물에 비해 폭 넓은 타겟 스펙트럼을 보여주었습니다. 이는 해당 약물의 인체에서의 독성 양태를 잘 대변하고 있으며, GPScreen™ 기술을 이용 시 동종의 신약 개발 후보 물질들 중에서 보다 안전한 약물을 선별 (drug prioritization) 함에 있어 매우 효과적으로 사용 가능합니다.
Figure 3. 독성 및 비독성 약물을 각각 GPScreen™ 한 후 타겟 스펙트럼 비교